Want to experience the greatest in board studying? Check out our interactive question bank podcast- the FIRST of its kind here: emrapidbombs.supercast.com
Author: Blake Briggs, MD
Objectives: discuss the pathophysiology, presentation, diagnosis, and management of acetaminophen overdose.
Introduction: acetaminophen (APAP, Tylenol, paracetamol) was introduced in 1955. Since then it has become the most popular analgesic in the world, and the most common cause of ingested toxicologic poisoning in the US. It is also the most common cause of acute liver failure in the Western hemisphere (50% of all cases). The popular quip regarding APAP is “the drug is extremely safe… until it’s not”.
–Less than 4000mg every 24 hours in adults (80 mg/kg in children) is the maximum limit.
Metabolism and Pathophysiology- Testable stuff on exams!
Normal APAP levels: APAP is metabolized to sulfate and glucuronide conjugates —> urinary excretion
Toxic levels of APAP: saturated safe pathways —> shunting of APAP to accessory pathway —> NAPQI (nasty little compound) made
NAPQI causes oxidative injury to the liver which is irreversible and can quickly progress to liver failure.
Accelerate your learning with our EM Question Bank Podcast
- Rapid learning
- Interactive questions and answers
- new episodes every week
- Become a valuable supporter
Stages of APAP Overdose
Stage I: < 24 hours. Nonspecific GI symptoms, possibly nausea/vomiting, lethargy, malaise. Some are asymptomatic. Laboratory studies are unremarkable.
Stage II: 24-72 hours. Evidence of hepatotoxicity. Elevated AST/ALT, bilirubin, PT/PTT. RUQ abdominal pain.
Stage III: 72-96 hours. Fulminant hepatic failure. Liver function tests peak during this time. All the signs and symptoms of acute liver failure with hepatic encephalopathy, jaundice, coagulopathy. AST/ALT will easily exceed >5-10,000, elevated PT/PTT and bilirubin, lactic acidosis, and renal failure.
(Please see our handout on “Hepatic Havoc: Acute liver failure” on our website for more details for management of patients with ALF).
Stage IV: >4 days. Either death occurs or complete resolution.
Diagnosis: serum acetaminophen level. Order on any patient with suspicion of overdose, either intentional or accidental.
Other tests to grab: salicylates (often taken in combo), UDS, CBC, CMP.
If the patient already exhibits signs of toxicity (Stage >1), order GGT, coagulation studies, lipase, ammonia.
The APAP level should be measured at 4 hours after time of ingestion, but often we are unsure of the exact time of ingestion. Therefore, if any suspicion for ingestion time being > 4 hours, immediately obtain APAP level upon arrival to ED and again at 4 hours following that.
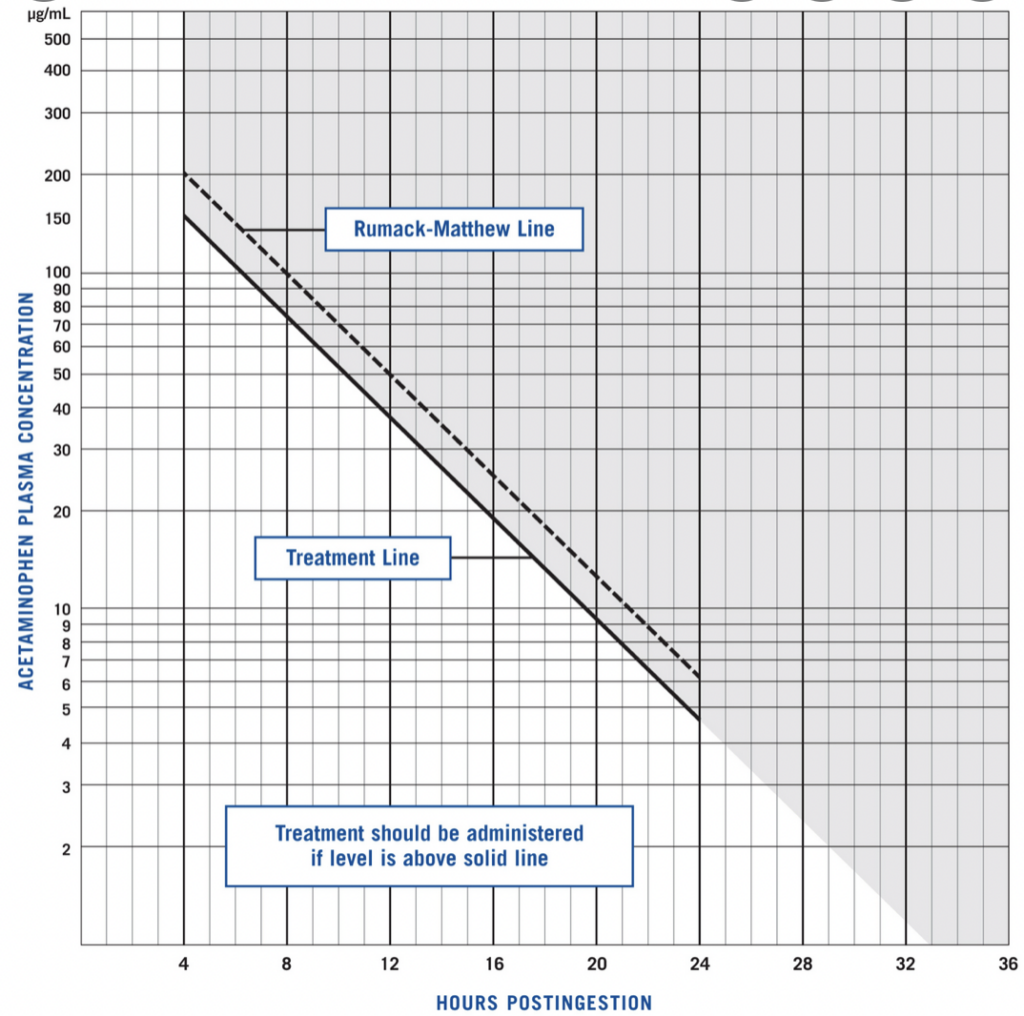
The APAP level should be evaluated using the Rumack-Matthew Nomogram (see to the right) to determine need for NAC therapy. Note: the nomogram is not applicable to chronic APAP overdoses.
Obtaining an initial serum APAP level will result in the following scenarios:
1. The initial APAP level at 4 hours since time of ingestion is negative, and patient is asymptomatic. You will want to observe the patient and remeasure at 4 hours again.
2. The initial APAP level at 4 hours since suspected time of ingestion is negative, but patient has signs of hepatic toxicity. Begin NAC therapy and remeasure at 4 hours.
3. The APAP level drawn on arrival with unknown time of ingestion is negative. If patient has signs of hepatic toxicity, start NAC and re-draw level at 4 hours. If patient is asymptomatic, observe patient for extended period and repeat in 4 hours
Management
Activated Charcoal: Patients presenting immediately (<3 hours probably but no hard line) after a potentially toxic ingestion of APAP could benefit from this. As always, concern most be given for airway protection first, but you should never intubate these patients just to give charcoal.
-Charcoal has been studied in APAP overdose and has been effective in reducing serum APAP concentration and reduce liver injury.
N-acetylcysteine (NAC) is very effective for preventing liver failure from APAP overdose. It is the only proven antidote for acetaminophen overdose.
Studies have repeatedly shown its superior profile in reducing serious hepatotoxicity (<4%) and death are extremely rare (<1%) if NAC is given <8 hours from overdose.
Mechanism is likely due to restoring glutathione stores.
There are no placebo trials (try passing that through the IRB). However, there is no downside to giving NAC. Even if it is given in late presentations and/or patients with liver failure from unknown cause, NAC can reduce cerebral edema and improve hepatic function.
While dosing errors are common when it comes to NAC, significant adverse events are rare.
-NAC can elevate the INR to <1.5 (interferes with lab analysis)
-NAC can cause vomiting (~30% of patients)
-NAC can cause anaphylaxis (10-20% of patients). This is obviously the most concerning side effect.
Prospective studies suggest that between 10-20% of patients treated with IV NAC have some type of hypersensitivity reaction. This is basically anaphylaxis that is not IgE-mediated. Reactions greatly vary in severity, and many are able to tolerate the infusion when it is restarted. But you need to be ready to manage anaphylaxis and not be surprised! So, let’s go through the scenarios here:
Patients with just flushing without pruritus or urticaria: no treatment needed. IV NAC can be continued.
Patients with urticaria, angioedema, or any respiratory symptoms: stop the infusion, give IM Epinephrine anaphylactic dose, as well as your usual antihistamine medications and steroids. The key here is you can restart the infusion at the prior rate once the urticaria resolves.
Patients with hypotension or other persistent systemic anaphylactic shock symptoms: stop the infusion, give IM epinephrine and the other meds, but do not restart NAC. You can give oral NAC though!
NAC can be given in pregnancy at the same dose.
IV route > Oral route. Both have been found to be basically equal but keep it simple and do IV in the majority of your patients.
There is a lot of detail and ongoing research regarding the duration of the NAC protocol- do not worry about this. Start NAC at the proper dose and coordinate with pharmacy regarding later dosages. Therapy typically lasts for 20-72 hours.
Disposition: patients can be admitted to floor (Medical-Psychiatric unit) if hemodynamically normal, even with NAC infusion. If signs of hepatotoxicity, or risk of anaphylaxis from NAC, admit to ICU.
Stopping NAC: Only if all the following are met- asymptomatic patient, APAP level nondetectable, serum transaminases have trended down or are normal.
References:
1. Chiew AL, Gluud C, Brok J, Buckley NA. Interventions for paracetamol (acetaminophen) overdose. Cochrane Database Syst Rev 2018; 2:CD003328.
2. Underhill TJ, Greene MK, Dove AF. A comparison of the efficacy of gastric lavage, ipecacuanha and activated charcoal in the emergency management of paracetamol overdose. Arch Emerg Med 1990; 7:148.
3. Spiller HA, Krenzelok EP, Grande GA, et al. A prospective evaluation of the effect of activated charcoal before oral N-acetylcysteine in acetaminophen overdose. Ann Emerg Med 1994; 23:519.
4. Smilkstein MJ, Bronstein AC, Linden C, et al. Acetaminophen overdose: a 48-hour intravenous N-acetylcysteine treatment protocol. Ann Emerg Med 1991; 20:1058.
5. Smilkstein MJ, Knapp GL, Kulig KW, Rumack BH. Efficacy of oral N-acetylcysteine in the treatment of acetaminophen overdose. Analysis of the national multicenter study (1976 to 1985). N Engl J Med 1988; 319:1557.
6. Prescott LF. Treatment of severe acetaminophen poisoning with intravenous acetylcysteine. Arch Intern Med 1981; 141:386.
7. Keays R, Harrison PM, Wendon JA, et al. Intravenous acetylcysteine in paracetamol induced fulminant hepatic failure: a prospective controlled trial. BMJ 1991; 303:1026.
8. Harrison PM, Wendon JA, Gimson AE, et al. Improvement by acetylcysteine of hemodynamics and oxygen transport in fulminant hepatic failure. N Engl J Med 1991; 324:1852.
9. Green JL, Heard KJ, Reynolds KM, Albert D. Oral and Intravenous Acetylcysteine for Treatment of Acetaminophen Toxicity: A Systematic Review and Meta-analysis. West J Emerg Med 2013; 14:218.
10. Bailey B, McGuigan MA. Management of anaphylactoid reactions to intravenous N-acetylcysteine. Ann Emerg Med 1998; 31:710.
11. Yip L, Dart RC. A 20-hour treatment for acute acetaminophen overdose. N Engl J Med 2003; 348:2471.
12. Whyte IM, Buckley NA, Reith DM, et al. Acetaminophen causes an increased International Normalized Ratio by reducing functional factor VII. Ther Drug Monit 2000; 22:742.
13. Rumack BH, Peterson RC, Koch GG, Amara IA. Acetaminophen overdose. 662 cases with evaluation of oral acetylcysteine treatment. Arch Intern Med 1981; 141:380.
14. Spooner, JB, Harvey, JG. Paracetamol overdose — Facts not misconceptions. Pharm J 1993; 251:706.
15. Bunchorntavakul C, Reddy KR. Acetaminophen-related hepatotoxicity. Clin Liver Dis 2013; 17:587.
16. Watson WA, Litovitz TL, Klein-Schwartz W, et al. 2003 annual report of the American Association of Poison Control Centers Toxic Exposure Surveillance System. Am J Emerg Med 2004; 22:335.
17. Lee WM. Acetaminophen and the U.S. Acute Liver Failure Study Group: lowering the risks of hepatic failure. Hepatology 2004; 40:6.
18. McGill MR, Jaeschke H. Metabolism and disposition of acetaminophen: recent advances in relation to hepatotoxicity and diagnosis. Pharm Res 2013; 30:2174.
19. Jollow DJ, Mitchell JR, Potter WZ, et al. Acetaminophen-induced hepatic necrosis. II. Role of covalent binding in vivo. J Pharmacol Exp Ther 1973; 187:195.
20. Singer AJ, Carracio TR, Mofenson HC. The temporal profile of increased transaminase levels in patients with acetaminophen-induced liver dysfunction. Ann Emerg Med 1995; 26:49.
21. Mazer M, Perrone J. Acetaminophen-induced nephrotoxicity: pathophysiology, clinical manifestations, and management. J Med Toxicol 2008; 4:2.



