Want to experience the greatest in board studying? Check out our interactive question bank podcast- the FIRST of its kind here: emrapidbombs.supercast.com
Author: Blake Briggs, MD
Objectives: define differences between upper and lower GI bleeds, decide need for acute intervention versus stable evaluation, describe pathologic causes of lower GI bleeds and algorithmic approach
Introduction: LGIB refers to GI bleeding below the ligament of Treitz. Mortality ranges from 2-4%.
A reminder about symptoms and presentations of UGIB vs LGIB (see handout on UGIB for more details). Remember: UGIB is the most common cause of GI bleeding overall!
Upper: from mouth to ligament of Treitz.
Lower: from the ligament of Treitz to anus.
Presentation:
UGIB: melena and hematemesis (either frank blood or coffee-ground material). 15% of UGIB have hematochezia. These patients are typically “sicker” appearing (more likely hemodynamically unstable, elevated BUN-Cr ratio).
LGIB: bright red blood if distal from left colon; dark/maroon bloody stool is typically found in the right colon. Melena is rare.
Accelerate your learning with our EM Question Bank Podcast
- Rapid learning
- Interactive questions and answers
- new episodes every week
- Become a valuable supporter
Hematochezia: maroon or bright red blood with or without clots. Presence of clots in stool dramatically raises chances of LGIB > UGIB.
Causes of acute LGIB in order: diverticulosis, anorectal causes (hemorrhoids, anal fissures, rectal ulcers), colorectal cancer, mesenteric ischemia, inflammatory bowel disease, angiodysplasia, infectious colitis
Hemodynamically stable or unstable?
-If any signs of volume loss, 2 large bore IVs are a priority- sooner rather than later!
-Examine for tachycardia (sign of hypovolemia and impending shock). Blood loss >15% causes orthostatic hypotension, blood loss >40% manifests in supine hypotension.
-If unstable, one must consider an UGIB source (as noted above).
It is important to note that the vast majority of all LGIB’s spontaneously stop bleeding (80%).
Diagnostic approach: CBC, type and screen, CMP, +/- Coags (only get the latter if patient has a history of anticoagulants, liver disease/alcohol abuse, inherited coagulopathy).
Hemoglobin is normal in the vast majority of patients during the initial 24 hours or more due to the patient losing whole blood. Do not trust the hemoglobin during acute blood loss! Giving IV fluids which dilute the hemoglobin cause a falsely lower reading in the first 24 hours.
The MCV should be normal in acute bleeding. A low MCV in the setting of anemia suggests chronic LGIB.
Obtain a detailed HPI (if hemodynamically stable and the patient is able to verbalize):
-how much bleeding, how long?
-is the bleeding painful?
-does the bleeding occur spontaneously, only with bowel movements, or only when wiping after a bowel mvt?
-pertinent positives: abdominal pain, nausea/vomiting, bowel movements
-stool: diarrhea? Formed, loose, or just frank blood? Bright red blood or dark and tarry? Clots? Painful?
Obtain PMH and social history (do not overlook this!)
-has this happened before? If so, was it treated? (up to 60% of previous LGIB/UGIB rebleed!)
-taking any anticoagulation?
-history of GI malignancy?
-history of AAA? Repaired?
-Prior GI surgeries?
-When was your last colonoscopy?
Perform a focused physical exam
-recheck the vitals!
-abdominal exam that demonstrates focal tenderness; guarding and rebound tenderness raise concern for peritoneal irritation (suggests perforation).
-rectal exam to determine presence of blood. Look for fissures, hemorrhoids, skin tags, fistulae, abscess, masses, condylomata.
What about the stool guaiac test or fecal occult blood test (FOBT)? FOBT has come under fire lately due to a lot of inaccuracies and lack of appreciable studies. There are high rates of false positives and negatives. One study showed they only change management 34% of the time, and in the majority of those patients 60% had endoscopy prior to the FOBT even being performed. Stool color is more sensitive for UGIB than FOBT. +FOBT does not rule in UGIB and negative FOBT does not rule out UGIB.
BUT, you still need to do a digital rectal exam. This can tell you if there is active rectal bleeding or blood in the rectal vault.
Initial resuscitation: If a patient is tachycardic, active bleeding present, and/or hypotensive with LGIB/UGIB symptoms or signs, there should be low threshold to transfuse early.
Regarding reversing anticoagulation, this is truly a case-by-case basis. Most approved guidelines favor continuing aspirin in patients with high-risk CAD. In general you should not discontinue dual-antiplatelet therapy in those with an acute coronary syndrome < 90 days or with a bare-metal stent placed <6 weeks or drug-eluting stents <6 months.
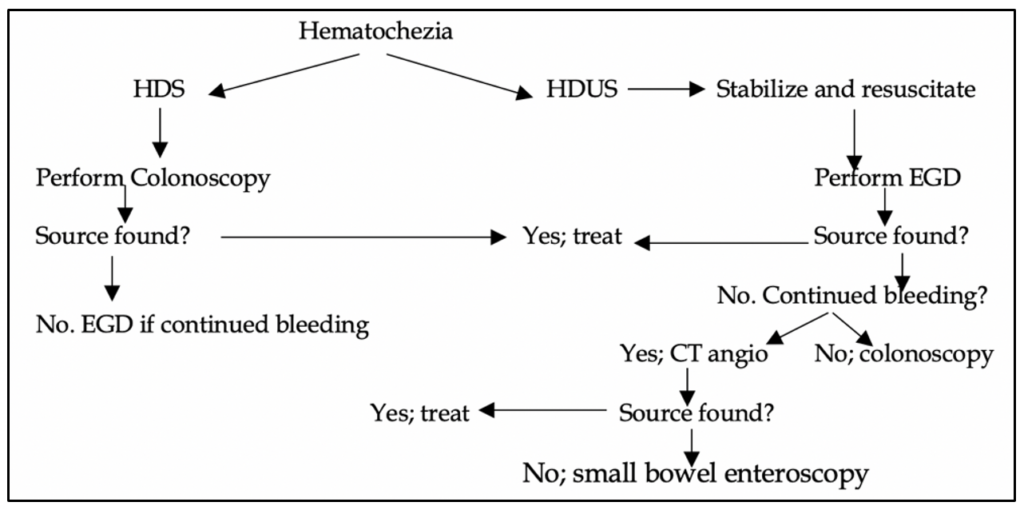
Details from Figure 1:
1) Rule out UGIB first.
2) Colonoscopy is the initial exam of choice in most LGIB patients. This does require bowel prep for best views.
3) Radiographic tests (CTA, radionuclide imaging, Mesenteric angiography all require active bleeding.
Of note, no randomized trials have demonstrated the advantage of any of the above tests over one another.
Colonoscopy: 45-90% successful in identifying a source. For best results, complete in <24 hours of presentation in patients.
CTA: 85% specific and 92% sensitive.
Patients with lower GI bleeding are a common ED complaint. CT angiography is widely available, faster, and less invasive than colonoscopy or endoscopy. In patients with ongoing, hemodynamically significant hematochezia, CT angiography is the initial diagnostic test of choice. However, the patient must have active bleeding for a positive scan. CT angiography without and with contrast of the abdomen and pelvis should be considered ONLY in the following situations in patients with suspected upper or lower GI bleeding:
- The patient is hypotensive with BP < 100 SBP or MAP <65.
- The patient’s hemoglobin is ≤ 7.
Details on specific causes:
Diverticulosis: most common cause of acute LGIB. Sac-like protrusion of colonic submucosa and mucosa, form a false diverticulum due to increased intraluminal pressures. These weak protrusions are prone to 2 major complications:
1) ruptured capillaries of the vasa recta which overlie the diverticulum dome, inciting acute bleeding.
2) fecal impaction within outpouching allow bacteria to accumulate and cause diverticulitis. In the western society, diverticula are found on the left in the vast majority of patients (in eastern culture it is on the right). Right side is prone to more bleeding.
Diagnosis: can be clinical suspicion, but CT abdomen/pelvis is much, much better. Colonoscopy is the gold standard.
Prevention of diverticulitis/losis: high fiber diet avoiding heavy meats and fats, stool softeners.
Treatment of diverticulosis: for acute diverticular bleeding, it is uncommon for patients to become unstable or to continue bleeding to warrant EGD.
Treatment of diverticulitis: Antibiotics (ciprofloxacin and metronidazole), admission if unstable or high fever, dehydration, not tolerating oral intake.
Mesenteric ischemia: see our handout on mesenteric ischemia for more details.
Angiodysplasia: excessively dilated, tortuous submucosal vessels which lack smooth muscle cells and are prone to rupture. Risk increases with age as blood vessels become weaker and less able to probably handle the high intraluminal pressures of the GI tract. Unlike diverticula, angiodysplasia is a venous bleed. Most common location of bleeding: cecum, right colon.
Weird associations: vWF deficiency and aortic stenosis (Heyde’s syndrome: vWF is “wasted” on the aortic valve due to macroangiopathic hemolysis, resulting in more bleeding in the distal GI tract due to high shear stress vessels in the intestines.
Treatment: colonoscopy with electrocoagulation, hemostasis, or argon laser coagulation
Anorectal disease: please see our handout on anorectal diseases for further details.
References
1. Mounsey AL, Halladay J, Sadiq TS. Hemorrhoids. Am Fam Physician 2011; 84:204.
1. Kelly SM, Sanowski RA, Foutch PG, et al. A prospective comparison of anoscopy and fiberendoscopy in detecting anal lesions. J Clin Gastroenterol 1986; 8:658.
2. Farrell JJ, Friedman LS. Review article: the management of lower gastrointestinal bleeding. Aliment Pharmacol Ther 2005; 21:1281.
3. Mortensen PB, Nøhr M, Møller-Petersen JF, Balslev I. The diagnostic value of serum urea/creatinine ratio in distinguishing between upper and lower gastrointestinal bleeding. A prospective study. Dan Med Bull 1994; 41:237.
4. Cappell MS, Friedel D. Initial management of acute upper gastrointestinal bleeding: from initial evaluation up to gastrointestinal endoscopy. Med Clin North Am 2008; 92:491.
5. Palmer ED. The vigorous diagnostic approach to upper-gastrointestinal tract hemorrhage. A 23-year prospective study of 1,4000 patients. JAMA 1969; 207:1477.
6. Richards RJ, Donica MB, Grayer D. Can the blood urea nitrogen/creatinine ratio distinguish upper from lower gastrointestinal bleeding? J Clin Gastroenterol 1990; 12:500.
7. Jensen DM, Machicado GA. Diagnosis and treatment of severe hematochezia. The role of urgent colonoscopy after purge. Gastroenterology 1988; 95:1569.
8. Jensen DM, Machicado GA, Jutabha R, Kovacs TO. Urgent colonoscopy for the diagnosis and treatment of severe diverticular hemorrhage. N Engl J Med 2000; 342:78.
9. Zuccaro G Jr. Management of the adult patient with acute lower gastrointestinal bleeding. American College of Gastroenterology. Practice Parameters Committee. Am J Gastroenterol 1998; 93:1202.
10. Baradarian R, Ramdhaney S, Chapalamadugu R, et al. Early intensive resuscitation of patients with upper gastrointestinal bleeding decreases mortality. Am J Gastroenterol 2004; 99:619.
11. Villanueva C, Colomo A, Bosch A, et al. Transfusion strategies for acute upper gastrointestinal bleeding. N Engl J Med 2013; 368:11.
12. Wu WC, Rathore SS, Wang Y, et al. Blood transfusion in elderly patients with acute myocardial infarction. N Engl J Med 2001; 345:1230.
13. ASGE Standards of Practice Committee, Acosta RD, Abraham NS, et al. The management of antithrombotic agents for patients undergoing GI endoscopy. Gastrointest Endosc 2016; 83:3.
14. Sung JJ, Lau JY, Ching JY, et al. Continuation of low-dose aspirin therapy in peptic ulcer bleeding: a randomized trial. Ann Intern Med 2010; 152:1.
15. Derogar M, Sandblom G, Lundell L, et al. Discontinuation of low-dose aspirin therapy after peptic ulcer bleeding increases risk of death and acute cardiovascular events. Clin Gastroenterol Hepatol 2013; 11:38.
16. Wilcox CM, Alexander LN, Cotsonis G. A prospective characterization of upper gastrointestinal hemorrhage presenting with hematochezia. Am J Gastroenterol 1997; 92:231.
17. Van Gossum A, Bourgeois F, Gay F, et al. Operative colonoscopic endoscopy. Acta Gastroenterol Belg 1992; 55:314.
18. Strate LL. Lower GI bleeding: epidemiology and diagnosis. Gastroenterol Clin North Am 2005; 34:643.
19. Dusold R, Burke K, Carpentier W, Dyck WP. The accuracy of technetium-99m-labeled red cell scintigraphy in localizing gastrointestinal bleeding. Am J Gastroenterol 1994; 89:345.
20. Scheffel H, Pfammatter T, Wildi S, et al. Acute gastrointestinal bleeding: detection of source and etiology with multi-detector-row CT. Eur Radiol 2007; 17:1555.



