Want to experience the greatest in board studying? Check out our interactive question bank podcast- the FIRST of its kind here: emrapidbombs.supercast.com
Fresh grounds: approach to UGIB in the ED
Author: Blake Briggs, MD
Peer Reviewer: Travis Smith, DO; Chris Musselwhite, MD
Objectives: define differences between upper and lower GI bleeds, decide need for acute intervention versus stable evaluation, describe pathologic causes of upper GI bleeds and an algorithmic approach to the UGIB patient.
Introduction: When approaching a patient who is bleeding from their GI tract, it is important to consider 1) their airway, 2) where the bleeding is coming from, which involves determining if it is from the Upper GI tract or Lower GI tract.
Upper: from mouth to ligament of Treitz.
Lower: from the ligament of Treitz to anus.
It is important to remember that UGIB are more common than LGIB.
Presentation:
Accelerate your learning with our EM Question Bank Podcast
- Rapid learning
- Interactive questions and answers
- new episodes every week
- Become a valuable supporter
UGIB: melena and hematemesis (either frank blood or coffee-ground material)
LGIB: hematochezia, rarely hematemesis.
90% of melena (dark tarry stool due to digested blood) originates from the upper GI tract (above the ligament of Treitz). However, hematochezia (bright red or maroon blood that is fresh) can come from an upper GI source if there is a massive bleed and blood is transiting the GI tract quickly. Of course, these patients would likely present hemodynamically unstable from the rapid blood loss.
These are the top 5 causes of UGIB in order of most prevalent: PUD, gastritis, varices, Mallory-Weiss tear, cancers.
1. Obtain an HPI (only if hemodynamically stable and the patient is able to verbalize)
-how much bleeding, how long?
-associated pertinent positives: abdominal pain, nausea/vomiting, bowel movements
-vomiting: How many times? is it bilious? Bloody? If bloody, is it frankly bloody or coffee-ground?
-stool: diarrhea? Formed, loose, or just frank blood? Bright red blood or dark and tarry? Clots? Painful?
2. Obtain PMH and social history (do not overlook this!)
-has this happened before? If so, was it treated? (60% of previous GI bleeders rebleed from the same lesion)
-use of NSAIDs or Goody’s/BC powder, aspirin, corticosteroids, anticoagulants?
-history of H. pylori diagnosis? Were they treated for it?
-history of GI malignancy, abdominal aortic aneurysm, or an aortic graft?
-use of alcohol? Look for signs of cirrhosis on exam.
3. Perform a focused physical exam
-recheck the vitals. Hypotension may be a late finding in young patients.
-rectal exam to determine presence of gross blood. +/- hemoccult assessment for occult blood but its reliability has been called into question. We do not believe in stool hemoccult cards, and this subject is discussed in the LGIB handout on the website.
4. Order these tests for every GI bleeder:
-CBC, CMP, type and screen/crossmatch
-Coagulation studies only in those with massive UGIB or history of anticoagulant usage, liver disease/ alcohol abuse, or oinherited coagulopathy.
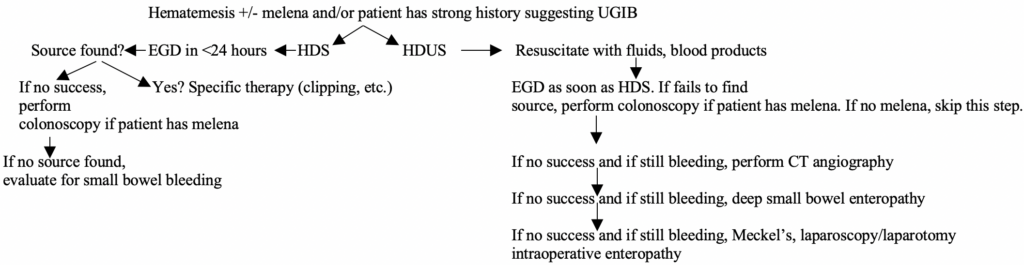
What about NG tubes? Minimal evidence supporting their usage. Many studies have agreed that they are about 66% accurate and even up to 15% of patients with an active UGIB will have a negative NG tube. They have been used in the past to lavage the stomach prior to EGD to improve visualization. They have not been shown to significantly help with this. We do not recommend their usage.
EGD: studies have shown it to be routinely 95% diagnostic, and 85% therapeutic.
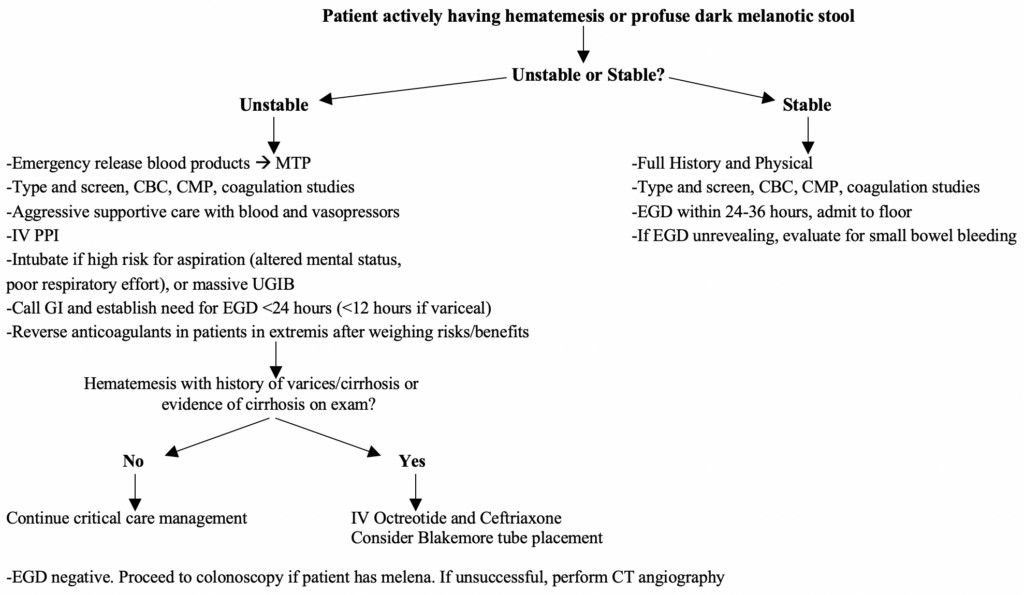
Where do we go from here? Well, here is the general flow chart in both unstable and stable UGIB patients:
Unstable UGIB critical details of management:
Large bore IV access is required. We do not hesitate to place ultrasound-guided 14-16g IVs if nursing has difficulty placing these blindly.
Airway assessment should be immediate. Intubation should be performed in those with ongoing, large volume hematemesis or altered respirations or mental status. However, elective endotracheal intubation has been associated with worse outcomes and should not be performed for all patients with hematemesis. Instead, intubate those who are high risk for aspiration, including those with altered mental status, worsening respiratory drive, or massive UGIB such as varices.
Resuscitation should not be delayed. Do not wait for a hemoglobin as this does not reflect acute blood loss.
Do not over-transfuse patients. Over-transfusion promotes more bleeding in variceal patients and may lead to higher mortality in all types of UGIB patients. Target hemoglobin should be <9 g/dL, <8 if history of CHF. Be quick to activate MTP in those requiring multiple blood products, as you should give FFP and platelets after every 4 units of PRBCs. Target platelet level >50,000. Give prothrombin complex concentrate if INR >2.
BUN increased at 24 hours compared to baseline is a predictor of poor outcomes. This is likely a reflection of poor resuscitation.
As the same with hemodynamically stable patients, everyone gets an IV PPI, and everyone will need endoscopy. However, the timeframe is much different. Patients can undergo EGD with thrombocytopenia, but do your best to properly manage platelet counts close to 50,000 and INR <2.5 as much as possible.
H2 blockers have not been shown to lower the rate of ulcer rebleeding. They should not be used. IV PPI (pantoprazole or esomeprazole are the only two IV options in the US). PPIs dramatically reduce rates of rebleeding and may promote hemostasis in patients with lesions other than ulcers, likely due to acid suppression with subsequent clot stabilization.
Prokinetics like erythromycin and metoclopramide can be given to improve gastric emptying and therefore enhance EGD visualization. This is reasonable to give and can be done 30-90 minutes prior to EGD.
In those hemodynamically unstable, EGD should be performed within 12 hours (if variceal bleeding), and <24 hours in others. The studies are controversial on this subject, as one study showed early EGD was actually associated with increased mortality. However, this is likely attributed to poor resuscitation. The emphasis should be on proper resuscitation in those with non-variceal UGIB, followed by EGD later.
Tranexamic acid: the famous antifibrinolytic that has been extensively studied in other areas of emergency medicine. It has not been shown to be beneficial here and should not be given to those with any type of GI bleeding.
Anticoagulation reversal: hotly debated and should be discussed case-by-case. Discussing with gastroenterology, patient and/or family, and the critical care team is necessary. For most patients, anticoagulation status should never delay EGD.
When is consulting gastroenterology for EGD the wrong decision? Aortoenteric fistulas. This is a general surgery/vascular surgery consult and gastroenterology cannot help in these cases. Any perforated viscous goes to the OR with surgery first.
Details on management of varices:
Esophageal varices cause painless esophageal bleeding with hematemesis due to portal hypertension. Increased pressure in the left gastric vein leads to blood pooling in the esophageal veins. They can burst and cause biblical levels of bleeding.
Presentation: cirrhotic patients with portal hypertension. They will present with frank hematemesis that began suddenly often with no precipitant vomiting. This is relentless hematemesis, very quickly necessitating airway protection.
Acute treatment: Airway protection —> resuscitation with large IVs —> IV octreotide*, IV PPI, IV ceftriaxone** —> EGD with banding or sclerotherapy —> if failed, balloon inflation in the esophagus and surgery discussed (e.g. TIPS). If there is any delay in performing EGD, discuss performing balloon inflation using a Blakemore or Minnesota tube.
*Octreotide works by reducing portal venous pressure. 50 mcg IV bolus, followed by infusion rate of 50 mcg/hour. We are honestly not big fans of it. It’s expensive and has no proven benefit in lowering mortality. Its role has been limited to cases where EGD is not immediately available as a means to stabilize bleeding before definitive therapy may be performed.
**Antibiotics in variceal bleeding: bacterial infections are associated with up to 20% of patients with cirrhosis who present with a UGIB. Up to 50% of patients will develop one while in the hospital. Both groups have increased mortality. What a crazy statistic. We recommend giving prophylactic antibiotics, as they may reduce rebleeding and infectious complications.
References:
1. Srygley FD, Gerardo CJ, Tran T, Fisher DA. Does this patient have a severe upper gastrointestinal bleed? JAMA 2012; 307:1072.
2. Cappell MS, Friedel D. Initial management of acute upper gastrointestinal bleeding: from initial evaluation up to gastrointestinal endoscopy. Med Clin North Am 2008; 92:491.
3. Jensen DM, Machicado GA. Diagnosis and treatment of severe hematochezia. The role of urgent colonoscopy after purge. Gastroenterology 1988; 95:1569.
4. Palmer ED. The vigorous diagnostic approach to upper-gastrointestinal tract hemorrhage. A 23-year prospective study of 1,4000 patients. JAMA 1969; 207:1477.
5. Richards RJ, Donica MB, Grayer D. Can the blood urea nitrogen/creatinine ratio distinguish upper from lower gastrointestinal bleeding? J Clin Gastroenterol 1990; 12:500.
6. Pallin DJ, Saltzman JR. Is nasogastric tube lavage in patients with acute upper GI bleeding indicated or antiquated? Gastrointest Endosc 2011; 74:981.
7. Ernst AA, Haynes ML, Nick TG, Weiss SJ. Usefulness of the blood urea nitrogen/creatinine ratio in gastrointestinal bleeding. Am J Emerg Med 1999; 17:70.
8. Huang ES, Karsan S, Kanwal F, et al. Impact of nasogastric lavage on outcomes in acute GI bleeding. Gastrointest Endosc 2011; 74:971.
9. Dorward S, Sreedharan A, Leontiadis GI, et al. Proton pump inhibitor treatment initiated prior to endoscopic diagnosis in upper gastrointestinal bleeding. Cochrane Database Syst Rev 2006; :CD005415.
10. Jutabha R, Jensen DM. Management of upper gastrointestinal bleeding in the patient with chronic liver disease. Med Clin North Am 1996; 80:1035.
11. Adang RP, Vismans JF, Talmon JL, et al. Appropriateness of indications for diagnostic upper gastrointestinal endoscopy: association with relevant endoscopic disease. Gastrointest Endosc 1995; 42:390.
12. Forrest JA, Finlayson ND, Shearman DJ. Endoscopy in gastrointestinal bleeding. Lancet 1974; 2:394.
13. Sarin N, Monga N, Adams PC. Time to endoscopy and outcomes in upper gastrointestinal bleeding. Can J Gastroenterol 2009; 23:489.

