Want to experience the greatest in board studying? Check out our interactive question bank podcast- the FIRST of its kind here: emrapidbombs.supercast.com
Authors: Travis Smith, DO, Charles Tindley OMS-4
Peer Reviewer: Blake Briggs, MD
Objectives: define ST-elevation myocardial infarction, review the pathology, diagnosis, treatment, and management.
Introduction: Acute Coronary Syndrome (ACS) is caused by disruption of coronary arteries by a plaque or thrombus leading to decreased perfusion and myocardial ischemia. Myocardial Infarction (MI) can present as an ST-segment elevation myocardial infarction (STEMI) or non-ST segment myocardial infarction (NSTEMI). Providers should be highly suspicious of ACS in patients presenting with chest discomfort and dyspnea as over 200,000 cases of STEMI are reported annually. That’s about 7.3 per 10,000 adult ED visits. This article will focus on those patients with STEMI. Please see our article on NSTEMI for that discussion.
Risk Factors: The five leading modifiable risk factors for coronary artery disease (CAD) are (1) hypertension, (2) diabetes, (3) obesity, (4) hypercholesterolemia, and (5) smoking. Over 90% of patients with CAD events, such as MI, have at least one risk factor. Family history is an independent risk factor for CAD. A first-degree relative prior to the age of 55 in males and 65 in females denotes a significant family history of premature atherosclerosis. Siblings of those with CAD have an increased risk of about 40%, while children of parents with premature CAD (Men <55 and Women <65) have an increased risk of 60-75%.

Pathophysiology: ACS typically occurs due to a rupture of a preexisting coronary lipid-laden plaque. This leads to the formation of a fibrin clot that can be partially or totally occlusive to the coronary vessel. Complete occlusion of the vessel leads to transmural cardiac ischemia in the territory of the blocked vessel. Clinically, transmural ischemia presents as an elevation of the ST segment on an EKG. This contrasts with a partial occlusion of the vessel which leads to subendocardial or non-transmural ischemia. Clinically, this presents signs and symptoms of ACS without ST-elevation on an ECG but with T-wave inversion, ST-depressions, and other non-specific ST-segment changes. Within minutes of infarction, there will be ECG changes.
Although most MI’s are caused by atherosclerosis, any condition that leads to a mismatch between oxygen demand and oxygen delivery can cause ischemic changes. These can include coronary emboli, thrombotic coronary artery disease (sickle cell anemia, polycythemia vera, thrombocytosis, disseminated intravascular coagulation), coronary vasculitis, coronary vasospasm, coronary dissection (think pregnant patient), coronary ostial occlusion, trauma, severe anemia, and myocardial profusion demand mismatch. The table to the right provides an overview of the classifications of MI’s.
Presentation: The most common symptom of STEMI is chest discomfort. In fact, chest pain and diaphoresis have the highest LR and PPV for STEMI. Chest discomfort is typically retrosternal but may also present as left parasternal, left precordial or across the anterior chest. Patients describe it as heavy, pressing, crushing, tightening, aching, or burning. Sites of radiation can include the left arm, right arm, shoulder, neck, jaw, teeth, epigastrium, and interscapular areas. Although the classic presentation of chest pressure with radiation to the jaw, left arm, or both arms is 96% specific, it is only about 11% sensitive.
Do not forget the atypical presentation of ACS. In nearly 20% of cases, MI’s are either painless (silent) or atypical. Elderly patients, women, and diabetics are particularly prone to atypical MI’s. Atypical MI presents with pain predominantly in the neck, jaw, ear, arm, or epigastrium. Other signs to look for STEMI: sudden dyspnea, weakness, diaphoresis, lightheadedness, hypotension, syncope, new arrhythmias, nausea, and vomiting.
Accelerate your learning with our EM Question Bank Podcast
- Rapid learning
- Interactive questions and answers
- new episodes every week
- Become a valuable supporter
In your initial evaluation, look for evidence of systemic hypoperfusion (hypotension, tachycardia, altered mental status, clammy/pale skin). This is concerning for cardiogenic shock associated with MI (5-10% of cases). For more details on cardiogenic shock and its management, see our other board bomb here.
Volume overload is not an expected finding as the onset is rapid. Rather, look for dyspnea, hypoxia, rales/wheezing. However, pulmonary congestion is an uncommon finding, only seen in <30% of patients.
TIMI score is a specific scoring system designed to determine risk of in-hospital mortality. But honestly, this is rarely used in the ED as the speed at which STEMI patients are addressed and given reperfusion therapy means they should (optimistically), be out of the ED no shorter than 45 minutes or less.
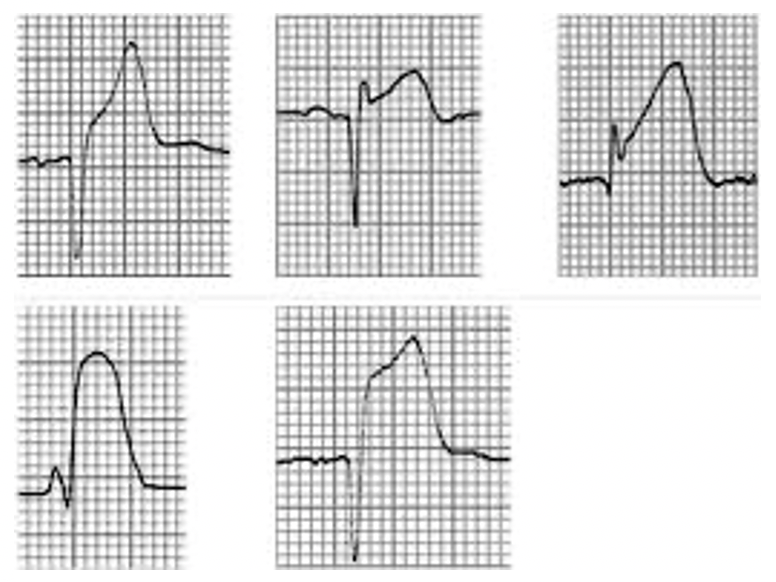
Diagnosis: Electrocardiogram and clinical symptoms are all that is needed to diagnose a STEMI. Biomarkers may be normal early on.
ECG Criteria: Remember the goal to get an ECG within 10 minutes of first encounter.
· ≥1mm ST-segment elevation in two contiguous chest or limb leads
· ≥2mm ST-segment elevation in men ≥ 40 years in leads V2-V3, ≥2.5mm in men < 40 years in leads V2-V3, and ≥1.5mm ST-segment elevation in women in lead V2-V3
Localization of ischemia can be determined by what ECG leads show ST- elevation.
· Anterior wall ischemia: two or more of the precordial leads (V1-V6); LAD V1-V4
· Anteroseptal ischemia: Lead V1-V4; Proximal LAD
· Apical or lateral ischemia: Leads aVL and I, V4 to V6: Distal/Diagonal LAD or LCx
· Inferior wall ischemia: Lead II, II, and aVF; 90% RCA, 10% LCx
Don’t forget about STEMI mimickers such as pericarditis (look for diffuse ST elevation without reciprocal T wave changes), myocarditis, hyperkalemia, LBBB, Brugada, Osborne waves in hypothermia, and early repolarization (look for J point notching). When reading EKGs for STE and STD, look at the J point. The J point is used to determine the magnitude of the ST segment elevation, and is compared to the isoelectric part of the tracing. In addition, AV block has been found in about 7% of cases and can result in bradycardia and low cardiac output. Have your pacer pads ready!
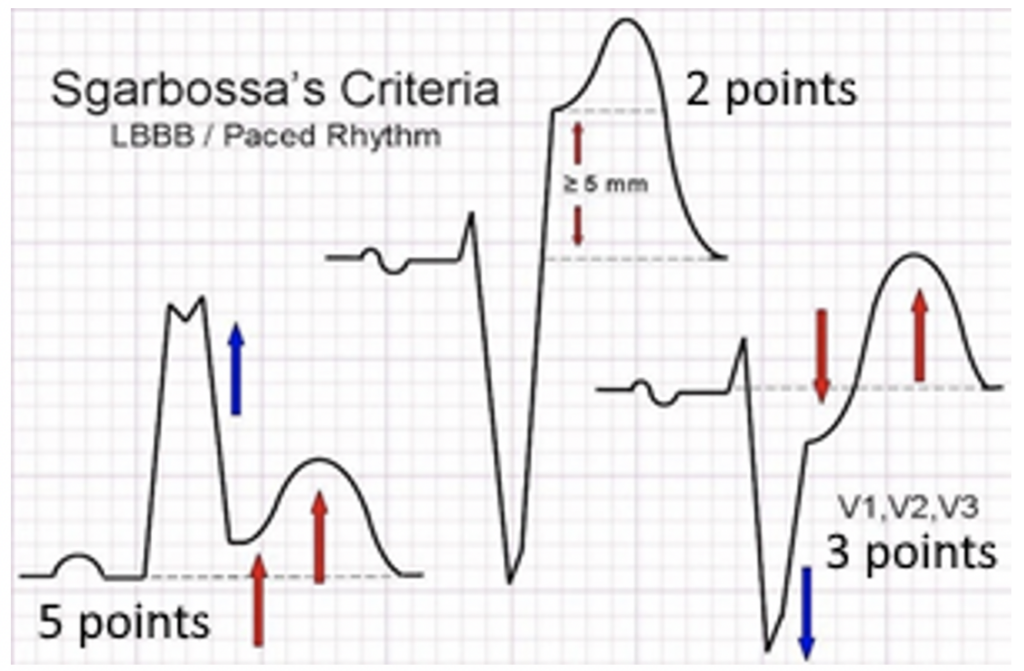
Speaking of LBBB, remember that it can obscure the ST-segment analysis and complicate ECG interpretation. The presence of a new LBBB is not a STEMI equivalent anymore, but when there is a new LBBB and other clinical and laboratory findings suggestive of an acute MI, it is associated with a high mortality. How can we tell? Thanks to the Sgarbossa criteria that was developed in the late 90s, a score of ≥ 3 has specificity of 98% but a sensitivity of 20%, increasing to a specificity of 100% and a sensitivity of 14% with score ≥5. There is now a modified Sgarbossa rule which highlights the criteria yielding only 2 points (seen in the middle of the photo on the right). Smith et al used an ST/S ratio of < -0.25 as a replacement for the absolute ST elevation of ≥5 mm. The most concerning finding is concordant STE. A simple way to apply the Sgarbossa is to look at each lead on the EKG for concordance. Each lead in a LBBB should be discordant or isoelectric. If there is concordance, stop and evaluate for Sgarbossa further.
Unique cases
STE in aVR is associated with >1 mm STD in other multiple leads may suggest LMCA stenosis or occlusion. However, more and more recently this has come under fire. It seems more commonly aVR elevation is due to subendocardial ischemia or evidence of triple vessel disease. In fact, tachycardia-related depression can even cause STE in lead aVR.
STE in aVR is not specific for LMCA, and the patient’s clinical condition should be taken into consideration.
Isolated Posterior MIs are rare: 3% of total MIs. More commonly, they are seen as an extension of an anterior infarction into much larger area involving the posterior. These will present as ST depression in leads V1-3 with a positive T wave. Get a posterior EKG (V7-9). This is easy. Move leads V4-6 to their mirror position on the back. ST elevation >0.5 mm in any posterior lead is diagnostic.
RV infarction is seen in up to 40% of inferior STEMIs. On a typical 12-lead, suspect when there is an inferior STEMI with ST elevation in V1 or V1 > V2, ST elevation in Lead III > II, or ST elevation in V1 with ST depression in V2. The latter is the most specific for RV infarct. Confirm RV infarction by performing a “Right sided EKG”. Place V1-V6 in a mirror image pattern on the R chest.
These patients are especially preload sensitive! These are the people you are avoiding giving Nitrates. Treat hypotension with IV fluids.
We cannot emphasize serial EKGs enough. We recommend getting one every 15-30 minutes for the first 1-2 hours as indicated, or anytime the patient’s pain changes. One report showed 8% of all STEMIs were identified on repeat EKG.
What about troponins?
Elevations in cardiac troponin is usually present in those presenting with a STEMI, however, if patients present soon enough (under an hour since symptom onset), the initial troponin can be negative. An elevation in cTnI above the 99th percentile of normal has is both sensitive and specific for myocardial damage. Troponins are detectable from 1 to 4 hours after the onset of an acute MI. They are maximally sensitive at 8 to 12 hours and peak at 10 to 24 hours.
Never wait for a positive laboratory test to proceed with a percutaneous coronary intervention (PCI) if ECG and clinical symptoms are highly suspicious for STEMI.
Imaging: In most places, a chest x-ray can be performed quickly not delaying transporting to the cath lab. If it delays transport, omit it. Yes, you should think about aortic dissection, but an abnormal CXR is only present in about 68% of cases so make sure you know that if you are highly suspicious, get a CTA chest. A bedside echo can help as studies in patients presenting to the ED with chest pain have found it to be very sensitive with regional wall motion abnormalities present in about 93% of cases with AMI, they are also found in 43% of those without AMI.
Check out our next handout on STEMI management as we wrap up this high yield topic here!
References:
1. Yusuf S, Hawken S, Ounpuu S, et al. Effect of potentially modifiable risk factors associated with myocardial infarction in 52 countries (the INTERHEART study): case-control study. Lancet 2004; 364:937.
2. Ward MJ, Kripalani S, Zhu Y, et al. Incidence of emergency department visits for ST-elevation myocardial infarction in a recent six-year period in the United States. Am J Cardiol. 2015;115(2):167-170. (Cross-sectional analysis of database; 1.5 million ED visits)
3. Glickman SW, Shofer FS, Wu MC, et al. Development and validation of a prioritization rule for obtaining an immediate 12-lead electrocardiogram in the emergency department to identify ST-elevation myocardial infarction. Am Heart J 2012; 163:372.
4. Goldman, Lee, et al. Goldman-Cecil Medicine. 25th ed., vol. 1, Elsevier Saunders, 2020.
5. Go AS, Barron HV, Rundle AC, et al. Bundle-branch block and in-hospital mortality in acute myocardial infarction. National Registry of Myocardial Infarction 2 Investigators. Ann Intern Med 1998; 129:690.
6. Fanaroff AC, Rymer JA, Goldstein SA, et al. Does This Patient With Chest Pain Have Acute Coronary Syndrome?: The Rational Clinical Examination Systematic Review. JAMA 2015; 314:1955.
7. Reddy KS, Satija A. The Framingham Heart Study: impact on the prevention and control of cardiovascular diseases in India. Prog Cardiovasc Dis 2010; 53:21.
8. Lemkes JS, Janssens GN, van der Hoeven NW, et al. Timing of revascularization in patients with transient ST-segment elevation myocardial infarction: a randomized clinical trial. Eur Heart J 2019; 40:283.
9. Dai X, Bumgarner J, Spangler A, et al. Acute ST-elevation myocardial infarction in patients hospitalized for noncardiac conditions. J Am Heart Assoc 2013; 2:e
10. Andreou C, Maniotis C, Koutouzis M. The Rise and Fall of Anticoagulation with Bivalirudin During Percutaneous Coronary Interventions: A Review Article. Cardiol Ther. 2017;6(1):1-12. doi:10.1007/s40119-017-0082-x
11. Fanaroff AC, Rymer JA, Goldstein SA, et al. Does this patient with chest pain have acute coronary syndrome? JAMA. 2015;314(18):1955.
12. Gokhroo RK, Ranwa BL, Kishor K, et al. Sweating: a specific predictor of ST-segment elevation myocardial infarction among the symptoms of acute coronary syndrome: Sweating In Myocardial Infarction (SWIMI) Study Group. Clin Cariol. 2016;39(2):90-95.
13. Sabia P, Afrookteh A, Touchstone DA, et al. Value of regional wall motion abnormality in the emergency room diagnosis of acute myocardial infarction. A prospective study using two-dimensional echocardiography. Circulation. 1991;84(3 Suppl):I85-I92.
14. A review of the Sgarbossa criteria is available at Calculated Decisions.
An online tool for the Sgarbossa criteria is available at: www.mdcalc.com/sgarbossas-criteria-mileft-bundle-branch-block.
15. Jain S, Ting HT, Bell M, et al. Utility of left bundle branch block as a diagnostic criterion for acute myocardial infarction. Am J Cardiol. 2011;107(8):1111-1116. (Retrospective; 892 patients)
16. Tabas JA, Rodriguez RM, Seligman HK, et al. Electrocardiographic criteria for detecting acute myocardial infarction in patients with left bundle branch block: a meta-analysis. Ann Emerg Med. 2008;52(4):329-336.
17. Smith SW, Dodd KW, Henry TD, et al. Diagnosis of ST-elevation myocardial infarction in the presence of left bundle branch block with the ST-elevation to S-wave ratio in a modified Sgarbossa rule. Ann Emerg Med. 2012;60(6):766-776.
18. O’Gara PT, Kushner FG, Ascheim DD, et al. 2013 ACCF/AHA guideline for the management of ST-elevation myocardial infarction: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Circulation. 2013;127(4):e362-e425.


